
Don’t Panic, We’ll Survive This Shutdown Too
The government is “shut down.” Media call this a “crisis.” A “crisis (with) no deal in sight,” says Fox News. Reuters says it's a “key risk to US stability.” But when I look around, I see business as usual -- families raise children, workers work, people play music ... The media act like government is the most important part of life. It isn't. Fortunately, most of life, and the best of life,…

FDA Chief Exposes Biden’s Pandemic Medical Censorship
The Trump-appointed commissioner of the Food and Drug Administration exposed the Biden administration on Fox News for seeking to censor potentially damning information about vaccines.

SCOTUS Throws Out Case Against Dangerous & Deadly Abortion Pill
The U.S. Supreme Court recently dismissed a lawsuit against the FDA's approval of an abortion drug, a decision that has sparked fear for women's health and safety among pro-life individuals and groups. Backed by Alliance Defending Freedom attorneys, healthcare professionals had voiced concerns over the lack of regulations and the drug's risky side effects, as well as the absence of a…
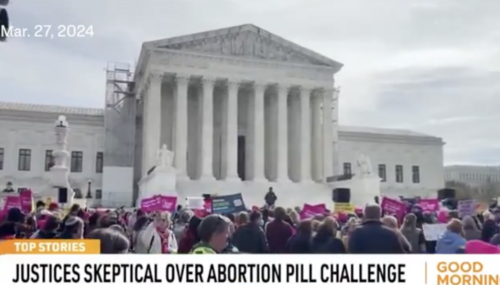
SCOTUS Hears Oral Arguments Against FDA's Approval of Abortion Pill
The U.S. Supreme Court heard the FDA v. Alliance for Hippocratic Medicine case regarding the safety of abortion pills. Supporters on both sides rallied outside the court. The Alliance for Hippocratic Medicine argued that the FDA had removed crucial safeguards for using and reporting on the side effects, resulting in harm to women. Protestors urged the FDA to ensure drugs help and don't harm…

Study Shows >60% of Abortions Are Done By Harmful Abortion Pill
The Guttmacher Institute, a pro-choice organization, has released a report indicating that over 60% of abortions in 2023 were conducted using abortion pills, a significant rise following the Supreme Court's decision to overturn Roe v. Wade in 2022. This method of abortion, involving a two-pill regimen, is considered four times more dangerous than surgical abortions and has led to a 500%…
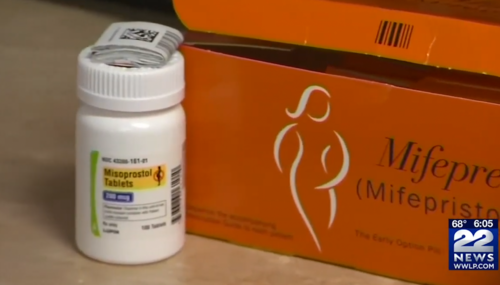
5th Circuit Rules FDA Must Restore Vital Safeguards for Abortion Pill
On August 16, the 5th Circuit Court of Appeals ruled that the FDA must restore crucial safeguards around chemical abortion drugs.

Supreme Court Rules Against Federal Ban On Abortion Pill
The news of this is shocking and heartbreaking for those in the pro-life movement.

PBS, NPR’s Free Ads for Abortion Pills: Click Here for More Info!
Who says the liberal media is anti-business? Taxpayer-assisted PBS and National Public Radio are eager to promote at least one product: Companies that mail the pregnancy-terminating pill mifepristone across state lines to states where abortion is being made illegal, in the wake of the Supreme Court’s Dobbs decision. And a pro-life group is suing claiming the FDA wrongly approved the…

VP Harris Lies In Order To Push Abortion Pill Support
Isn’t it great when the so-called “leaders” of our country lie to us?

MORE CENSORSHIP? FDA Commissioners Push Fight Against Misinfo
Food and Drug Administration commissioners have reportedly called for the agency to find a way to fight so-called “misinformation” online. If history is any indicator, this just means the agency is calling for more censorship.

FDA Warns Puberty Blockers Cause Vision Loss, Brain Swelling
The leftist prelates of transgenderism have been insisting that the puberty blockers used to ‘transition’ children are ‘completely reversible’ and ‘safe’. Of course, this is a lie, and the FDA has confirmed that by warning of potential vision loss and brain swelling for children that take puberty blockers, as …

Does the Constitution Need to Be Amended?
This Fourth of July, watching people fight over what the Constitution means, I ask people, if you could change the Constitution, what would you change? “The forefathers knew what they were doing,” said one woman. But the Constitution originally accepted slavery. It's good that we can amend it. So what should we change? “Add a balanced budget amendment,” suggests Glenn Beck.
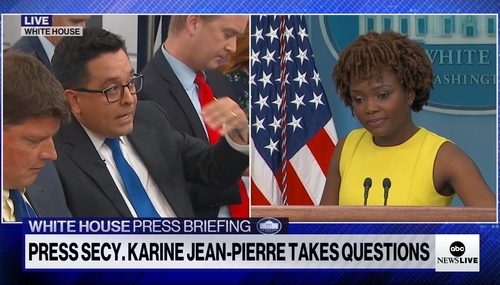
Try Harder: Jean-Pierre Walks into Another Buzzsaw of Doocy, O’Keefe
For the second day in a row on Thursday, White House Press Secretary Karine Jean-Pierre struggled to offer even cogent spin for the Biden administration on gun control and their months-long delay in responding to the baby formula crisis. Once again, it was Fox’s Peter Doocy and CBS’s Ed O’Keefe that most agitated Jean-Pierre.
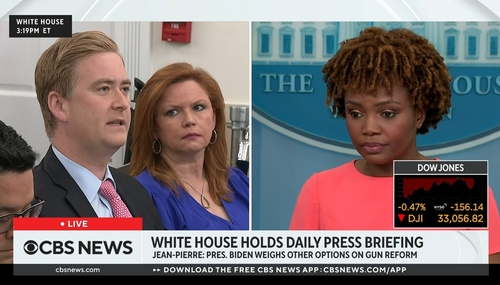
TRAINWRECK: WH Journos Torch Jean-Pierre on Formula Crisis, Inflation
White House Press Secretary Karine Jean-Pierre suffered the first of what could be many terrible, horrible, no good, very bad days as reporters from conservative and liberal outlets as well as the front and back of the room hammered away with questions about the baby formula shortage, inflation, and presidential leadership. For Jean-Pierre, she had little in the way of answers besides lengthy…



